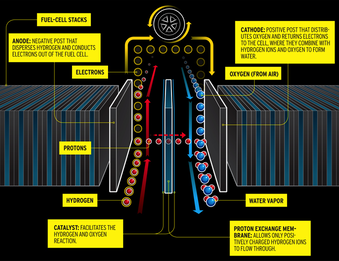
Car and Driver is the latest enthusiast rag to hop onto the hydrogen bandwagon with their article “What the H?” in this month’s issue. I wrote this letter to the editor but, since I doubt it will be printed, I’m reproducing it here.
Editors,
I’m sorry to see that you’ve bought into the hydrogen hype (“What the H”, Jan 2012). Hydrogen fuel cells make for nice demonstration projects, but will never be a meaningful part of the national transportation infrastructure.
Why? First, there are no hydrogen wells. You have to create it, whether from water electrolysis, biomass gasification, or natural gas reformation. According to the Second Law of Thermodynamics, that’s always going to take significantly more energy than you get out of burning it in a fuel cell. In this sense, hydrogen is just a bulky, expensive, and occasionally-dangerous battery.
Second, hydrogen is incredibly hard to work with. It can’t be transported or stored easily. Standard tools, fittings, tanks, and materials become brittle or leaky. Yes, aerospace companies have solved these problems for rocket engines, but not with parts you can buy at Pep Boys.
Finally, hydrogen is the opposite of dense. Whether as a liquid or a pressurized gas, a hydrogen tank contains only a fraction of the potential energy represented by an equal-size tank filled with liquid hydrocarbons. That’s a fundamental physical limit, and can’t be improved by smart engineering.
If you want to burn hydrogen in your car, the best way is to attach your hydrogen atoms to carbon atoms… and make gasoline.
Thanks for the otherwise-great issue!
Stephen